Article-HIV Assembly
Roles of Gag-RNA interactions in HIV-1 virus assembly deciphered by single-molecule localization microscopy
Abstract:
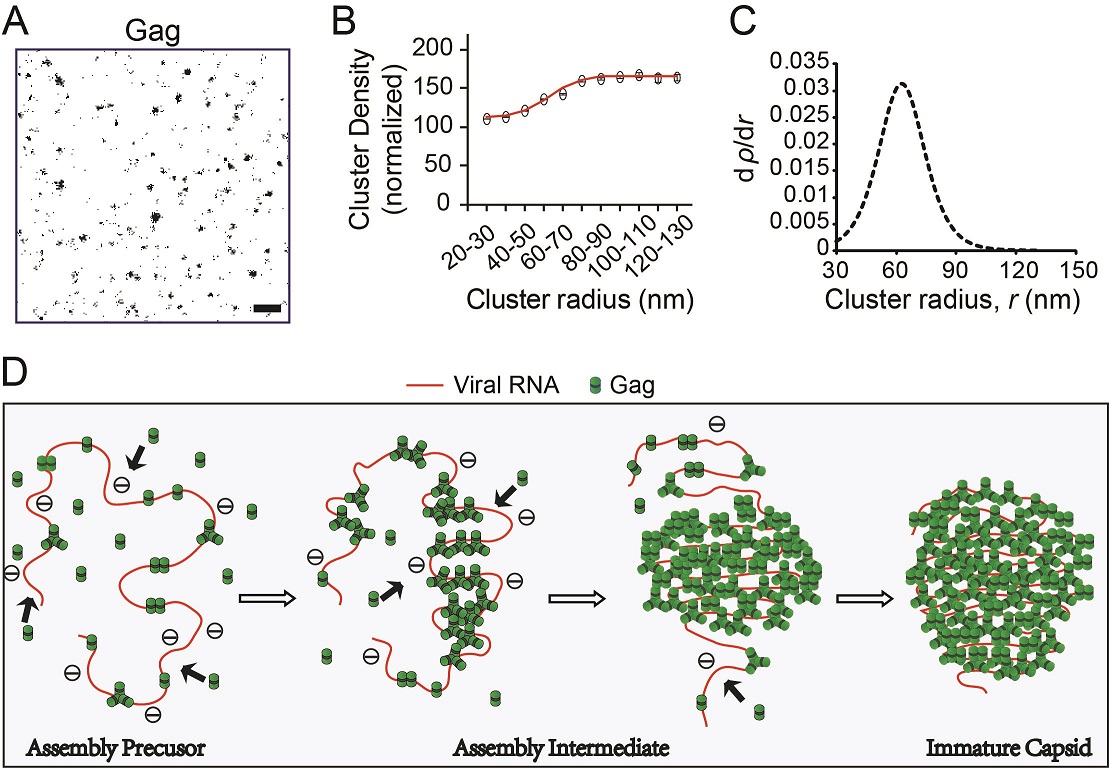
During HIV-1 assembly, the retroviral structural protein Gag forms an immature capsid, containing thousands of Gag molecules, at the plasma membrane (PM). Interactions between Gag nucleocapsid (NC) and viral RNA (vRNA) are thought to drive assembly, but the exact roles of these interactions have remained poorly understood. Since previous studies have shown that Gag dimer- or trimer-forming mutants (GagZiL) lacking an NC domain can form immature capsids independent of RNA binding, it is often hypothesized that vRNA drives Gag assembly by inducing Gag to form low-ordered multimers, but is dispensable for subsequent assembly. In this study, we examined the role of vRNA in HIV-1 assembly by characterizing the distribution and mobility of Gag and Gag NC mutants at the PM using photoactivated localization microscopy (PALM) and single-particle tracking PALM (spt-PALM). We showed that both Gag and GagZiL assembly involve a similar basic assembly unit, as expected. Unexpectedly, the two proteins underwent different subsequent assembly pathways, with Gag cluster density increasing asymptotically, while GagZiL cluster density increased linearly. Additionally, the directed movement of Gag, but not GagZiL, was maintained at a constant speed, suggesting that the two proteins experience different external driving forces. Assembly was abolished when Gag was rendered monomeric by NC deletion. Collectively, these results suggest that, beyond inducing Gag to form low-ordered multimer basic assembly units, vRNA is essential in scaffolding and maintaining the stability of the subsequent assembly process. This finding should advance the current understanding of HIV-1 and, potentially, other retroviruses.
See full-text: https://doi.org/10.1073/pnas.1805728115
Yachen Ying PUBLICATION
Single-molecule localization microscopy HIV assembly Gag-RNA interaction